MICRODIGEST, a Micro-device for Human Gastrointestinal Tract Simulation
July 22, 2022
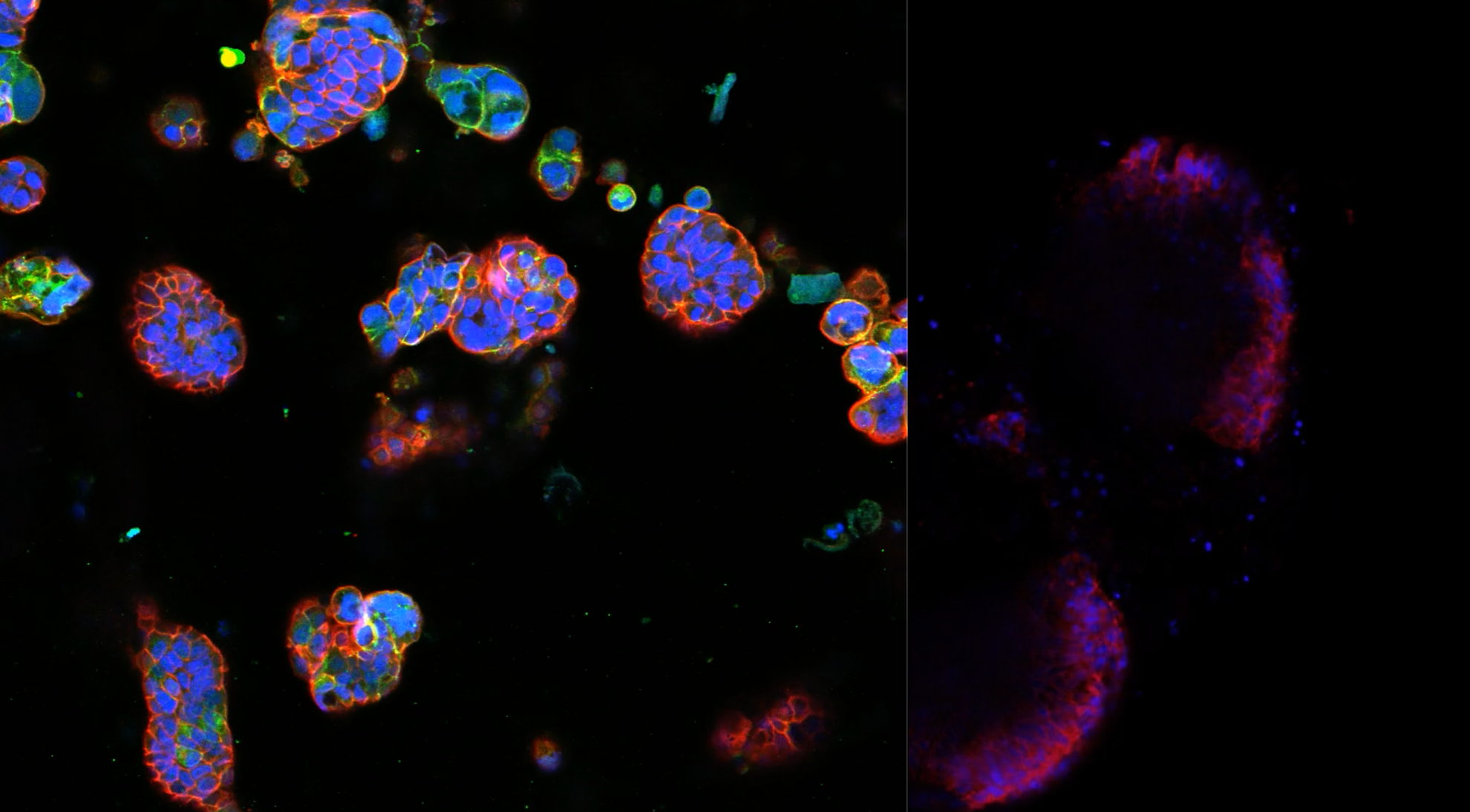
Orally administered bioactive compounds (nutraceuticals, pharmaceuticals, among others) show their effects only after gastrointestinal digestion and intestinal absorption. GI digestion by gastric acid and/or enzymatic degradation may deactivate oral administered active compounds. Encapsulation is often required, using nanoparticles, nanocapsules, micelles, etc., to protect the bioactive compound.
In the MICRODIGEST project, INL researchers developed a tool to evaluate the safety and efficacy of new drugs or foods. The modular sensorised platform uses microfluidics to simulate human digestion (oral, gastric, and intestinal phases) and intestinal absorption in an automated and miniaturised manner. This allows for reducing sample size, increasing experimental throughput and potentially reducing costly failures in future clinical trials.
During the project, different coatings of the PDMS were tested to reduce the absorption of small molecules, since hydrophobic and fluorescent molecules tend to diffuse into the PDMS reducing their concentration in solution and consequently affecting the accuracy and reliability of the assay. Researchers combined and strengthened the knowledge of different research teams at INL: cell culture, microfluidics and microfabrication. It was its interdisciplinarity that made this project obtain great results.